Key points
- As Australia's and Southeast Asia's demographics shift towards larger, ageing populations, demand for healthcare will continue to increase to 2040.
- This will result in greater demand for skilled medical workers, high-value pharmaceuticals, and quality health infrastructure investment.
- Australian healthcare providers are well placed to meet some of this demand; there will also be opportunities for two-way movement of skilled workers.
- Australia's development cooperation program will continue to support health outcomes in Southeast Asia.
Sector overview
Southeast Asia's fast-growing population will create a major increase in demand for healthcare services and infrastructure to 2040. This will put significant pressure on health authorities across the region. New spending needed on health infrastructure is estimated at up to US$39.1 billion per year on average to 2030, with Indonesia accounting for around 40 per cent of this.187 Constraints on public budgets will see demand continue to outstrip government capacity, and the private sector will be key to filling the gap.
There will be more than 100 million people aged 65 and older in Southeast Asia and over 6 million in Australia by 2040,188 necessitating greater government healthcare spending. With health expenditure estimated to be three to five times higher for older people, this will increase demand for specialised aged care.189
As the region grows more prosperous, there will be growing demand for higher-quality medical goods and services. Private spending on preventative care will continue to rise. According to industry submissions to this strategy, consumers in the region are moving from healthcare to wellbeing care, resulting in rapid growth in the market for preventative treatments, including complementary medicines. Southeast Asia's market for supplements is projected to be US$10.6 billion by 2026.190
Digital health will continue to have a transformational impact in Southeast Asia to 2040, as advances in digital technology provide low-cost ways to improve health outcomes. Southeast Asian governments are building the architecture needed to integrate digital solutions into healthcare systems, particularly to deliver cost-effective care for underserved rural and remote communities across the region. Digital health will also open opportunities for health services trade without the need for large physical in-market presence, as the Medical Technology Association of Australia highlighted in its submission.
As demographics and trends shift towards greater uptake of health goods and services, there will be further trade and investment opportunities for Australian and Southeast Asian businesses in healthcare. Health goods and services already represent a notable component of trade between Australia and Southeast Asia, with Australian medical-related goods exports to and imports from Southeast Asia worth A$520 million and $1 billion, respectively, in 2022.191
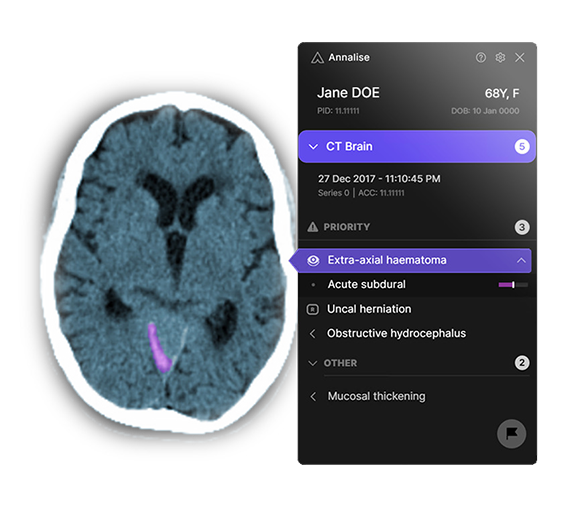
There will also be opportunities for Australian healthcare providers in remote monitoring and healthcare management software, including hospital information systems and electronic medical records, cloud services, data security and management, artificial intelligence, and data analytics and diagnostics (see Annalise.ai case study). In submissions to this strategy, medical providers noted the high take-up of technology across Asia was driving growth in virtual care. This will combine with fiscal and other capacity constraints to drive demand for cost-efficient and cost-effective healthcare. McKinsey estimates that, by 2025, over 75 per cent of the value of healthcare in Asia will be driven by digitalisation of care delivery.192
Case study: Annalise.ai improving clinical decisions and patient outcomes in Southeast Asia through artificial intelligence
Annalise.ai is a medical imaging artificial intelligence (AI) company formed as a joint venture between I-MED Radiology Network, Australia's largest diagnostic imaging services provider, and harrison.ai, a leading health tech company.
Annalise.ai's solutions are built using leading-edge AI technology to analyse radiology images and detect clinical findings, empowering clinicians to make faster and more accurate diagnostic decisions, helping lower treatment costs and improving patient outcomes.
Southeast Asia plays a significant role in Annalise.ai's growth ambitions, though there are challenges to overcome. Lakshmi Gudapakkam, CEO of Annalise.ai, said, 'The regional demand to improve efficiency and patient care through advanced technologies is a perfect fit for Annalise.ai's vision and strategy.'
'In the healthcare industry, the use of AI is still in its early stages, bringing forth several challenges. One prominent challenge is the need for a robust and scalable digital IT infrastructure. Another challenge is the change management involved in the adoption of any new technology, including workforce training and investments,' said Mr Gudapakkam.
Annalise.ai's solutions are already seeing success in the region. It has signed agreements with the Ramsay Sime Darby Group and the Gleneagles Hospital Kuala Lumpur in Malaysia, and the Hoan My Saigon Hospital in Vietnam for collaboration.
Southeast Asia's tech-capable workforce is also playing an important role in Annalise.ai's operations, with around half of its 300-plus global workforce located in Southeast Asia. In addition, Annalise.ai continues to focus its philanthropic efforts in the region. For example, it has been working with industry partners ASIF and Fujifilm to deliver a portable X-ray machine equipped with its AI technology, Annalise CXR Edge, at Thanh An Island in Vietnam, making significant improvements in people's life.
Partnerships between Southeast Asian and Australian companies help boost understanding of local markets and build resilient business relationships. For example, working closely with local joint venture partners helps navigate limits on foreign ownership of health facilities in most markets, and also assists Australian companies to navigate complex local market dynamics (see Aspen Medical case study). Australian healthcare providers can also take advantage of a range of tax and other incentives to establish private hospitals or public-private partnerships in the region.
Submissions to this strategy further highlighted the importance of Austrade's in-market knowledge, particularly to increase understanding of key purchasers, distributors and procurement processes for health and medical products, and to facilitate introductions. Consultations also highlighted the need to continue building recognition of Australia's health capabilities in the market, particularly with strong competition from companies from the United States, Japan, China, the European Union and the Republic of Korea.
The aged care and geriatric medicine sectors offer particular opportunities. Current care patterns in the region often see older people cared for by family – usually women – in the home, limiting demand for nursing homes, retirement villages and geriatric clinics.193 This unpaid care load means gender gaps in paid work and compounds gender inequalities. Investment in care infrastructure will help to redistribute the burden of care and create quality job opportunities, particularly for women. In the short term, demand is likely to be strongest for home-based care before shifting towards rising demand for facility-based care. In Singapore and Thailand, smart technologies are expected to support the delivery of cost-effective and high-quality care for large elderly populations, including assisted living, telehealth and remote monitoring technologies.194
Australian providers could help meet this need through a range of services, including training in elderly care, training in the differentiated geriatric care needs of men and women, specialist in-home geriatric services, and development and management of aged care facilities.
As Australia continues to age as a society, with the number of Australians over 85 expected to double in the next two decades to over 1 million,195 demand for skilled medical workers and aged care workers in Australia will continue to increase to 2040. There are clear opportunities for workers from Southeast Asia to bridge skills shortages in Australia, including for short periods of time.
“Southeast Asia is a big part of our future, as a base for our global logistics, supply chain and information technology services …” (Cochlear)
Southeast Asia is both a major importer and exporter of medical devices and equipment, presenting opportunities to export Australian-made products, invest in medical manufacturing to serve regional and global markets, and grow imports of medical equipment produced in Southeast Asia to ensure resilient health supply chains for Australia. As detailed in the case study below, Cochlear has engaged in Southeast Asia for over a decade.
Case study: Aspen Medical helping to improve healthcare in Indonesia
There is huge demand for new and upgraded health infrastructure across Indonesia to respond to rising consumer expectations for better healthcare experiences.
In 2020, Aspen Medical formed a partnership, formerly called Sanusa Medika, with Indonesian state-owned enterprise PT Jasa Sarana to build hospitals and community healthcare clinics in West Java.
With high demand for services for women, children and reproductive healthcare, these facilities will see opportunities to improve healthcare outcomes for women and children. Australian Government support has played an important role, with the Katalis partnership program under the Indonesia–Australia Comprehensive Economic Partnership Agreement funding feasibility studies on the first two hospital projects.
According to Aspen Medical Indonesia CEO, Dr Andrew Rochford, a long-term view and an appreciation of the local business culture is key.
'These markets are not without risk, but Indonesia is liberalising and if you take the time to build personal relationships and work through some unexpected challenges, Indonesia is an exceptionally welcoming market,' said Dr Rochford. 'We have been lucky to have the support of the Australian Ambassador to Indonesia and the Katalis program.'
While regulatory uncertainty and changes can create challenges to doing business in Indonesia, Aspen Medical has drawn on the knowledge and experience of its local partner and its own willingness to learn the local business culture and practices to navigate through these events.
Aspen Medical is continuing to grow its presence in Indonesia, including partnering with Bundamedik Healthcare System to train and empower local nurses and develop programs to improve women's health in east Indonesia.
Case study: Cochlear expands its presence in Southeast Asia
Established in 1981, Cochlear is a global leader in hearing-implant devices. Its technologies have transformed the lives of more than 700,000 people globally, including 9,000 people in Southeast Asia. The region is a growing support base for the company's global business activities.
Initial market entry into Southeast Asia was through partnering with distributors in each country. Cochlear was the first company to offer Cochlear implants, so the entry strategy was focused on building the market by training surgeons, building awareness of the indications and benefits of Cochlear implants, and – very importantly – educating governments. This is an inherently long-term approach requiring time and capital. The path for care, the expertise and infrastructure all had to be developed from the ground up. As markets grew, however, Cochlear opened offices in countries and slowly built its own employee base. These employees worked alongside its distributors, including in Thailand, Singapore and Malaysia. Each country has different practices and healthcare systems, so strategies had to be specific to each market.
In 2012, Cochlear began building an ICT support unit in Malaysia to provide a global help desk, systems architecture and design, and database support. The company expanded in 2016, centralising global repair and maintenance activities in Kuala Lumpur. Malaysia was selected due to its deep pools of skilled workers and business-friendly climate. Today, its Malaysia facility is the third-largest site in the Cochlear network, with locally recruited technicians manufacturing sound processors for Cochlear hearing-implant systems for customers around the world.
Cochlear's investment in the region also involves building medical skills. At the Cochlear Training and Experience Centre in Jakarta, Cochlear's objective is to share knowledge with local audiologists. Cochlear CEO and President, Dig Howitt, sees great potential in Southeast Asia.
'Cochlear is a global company and Southeast Asia is a big part of our future, as a base for our global logistics, supply chain and information technology services and a growing market for hearing implants,' said Mr Howitt.
As a proudly Australian company, Cochlear has worked closely with Austrade's regional network to make connections in local markets and recommends that others looking to the region do the same.
Cochlear's confidence in the manufacturing capability and international competitiveness of the region saw Cochlear commit in late 2022 to a RM30 million expansion at its Malaysian site. This will support growing demand for Cochlear's hearing implants globally. The new facility will be powered 100 per cent by renewable energy, supporting Malaysia and Cochlear to reach their net zero targets by 2050.
Demand for high-value pharmaceuticals and biotechnology products, including vaccines, will continue to grow across the region. Local partnerships will be important, as countries seek to expand their own manufacturing capabilities. Moderna's investment in mRNA manufacturing capability and research and development in Australia will also present opportunities to collaborate with the region on new vaccines targeted at regional health issues. There will also be potential to partner with companies and research institutions to accelerate research and development and commercialisation.
In addition to the opportunities these trends will offer businesses, government engagement will be fundamental to supporting strong and resilient health outcomes in the region. Australia is supporting universal health coverage in Southeast Asia through the Advance Universal Health Coverage Multi-Donor Trust Fund with the World Bank, by strengthening health financing and health service delivery systems. Australia's National Health and Medical Research Council has also funded 103 research grants worth A$95 million from 2013 to 2022 for research involving partners in Southeast Asia on issues ranging from eliminating tropical disease to dementia care.196
At a regional level, ASEAN member states and international partners have established the Asia eHealth Information Network to assist Asian countries to develop and implement national digital health strategies. Future priorities include standardising and strengthening regulatory frameworks; improving digital literacy, capacity and infrastructure; standardising the quality and interoperability of digital applications; and enriching regional and global collaboration.
Australia's development cooperation programs are already supporting strong and resilient health systems in Southeast Asia. Through Partnerships for a Healthy Region,197 the Australian Government is investing A$620 million in the health of Southeast Asia (and the Pacific), including strategic partnerships and projects aimed at preparing for and responding to infectious disease outbreaks; the detection and early treatment of non-communicable diseases; and supporting health promotion, mental health awareness and suicide prevention. The partnership also supports the development and introduction of novel vaccines, drugs, diagnostics and vector control technologies to the region. It works to expand government agency partnerships, including the work of Australia's Therapeutic Goods Administration (TGA) in strengthening the capacity of national regulatory authorities, and CSIRO's support in areas such as laboratory strengthening, biomedical manufacturing and digital health.
Pathways to 2040
In addition to the cross-cutting recommendations outlined in Chapter 2, which will have a broad economic impact, this chapter has additional specific recommendations on healthcare.
Remove blockages
The wide range of national regulatory frameworks in the region is a challenge. Pharmaceutical companies and regulated medical device manufacturers need to lodge a product application with each national regulatory authority for the product to be available on the market, leading to lengthy regulatory approvals that delay market access for many products in Southeast Asia.198 Australia's TGA, Southeast Asian national regulatory authorities and regional mechanisms such as ASEAN's Joint Assessment Procedure for Pharmaceutical Products are working to increase mutual recognition, cooperation and reliance among Southeast Asian markets so products approved in one Southeast Asian market can access several markets.
Australia should also explore ways to improve recognition arrangements for TGA-approved products by Southeast Asian national regulatory authorities to allow for speedier access to regional markets, including via memorandums of understanding to support exchange of information and documentation between national regulatory authorities.
Recommendation
- Australian Government to work with the medical industry to expedite efforts towards recognition or reliance arrangements for Australian-made, TGA-approved medical devices and pharmaceuticals in Southeast Asia.
Inconsistencies in labelling requirements and halal certifications pose challenges, particularly for complementary medicines. Businesses need to factor in the cost of country-specific labelling rules and obtaining halal certifications for different markets. Australian complementary medicine, health supplement and pharmaceutical producers also face strong competition from manufacturers in Southeast Asia and nearby regions that limit opportunities to compete in the low-cost segment of the market. The Australian Government will need to continue to work with industry to address market barriers in the region, including through the whole-of-government Non-Tariff Barriers Action Plan.
Recommendation
- Australian Government to work with industry and regulators to identify and address high-priority non-tariff barriers in the health sector.
Build capability
World Health Organization analysis shows Southeast Asia faces the world's largest needs-based shortage of nurses and midwives.199 Ratios of doctors in Southeast Asia also sit below the OECD's average in many markets.200 Australia's own workforce needs will also be significant.201
Building workforce capability will be key to uptake of digital and advanced medical technologies in the region. Collaborations between training providers, clinical societies, and digital and medical technology companies could lift both the region's workforce capability and further new medical technologies and treatments, improving health outcomes and opening commercial opportunities.
Australian institutions and training providers could help train a health workforce to the benefit of Australia and Southeast Asia, involving short-term placements in Australia. Australian education providers would play a key role, training medical practitioners to attain relevant qualifications in-country, either through their own campuses or in partnership with regional institutions, or in Australia and working with medical centres to meet clinical placement requirements.
Monash University Malaysia is a potential model, with doctors and pharmacists at its Kuala Lumpur campus trained to Australian standards and able to conduct their clinical placements in Australia. Such a scheme could align with reforms being considered by federal, state and territory health ministers in response to the interim report of the independent review conducted by Robyn Kruk AO, which recommends strategic partnerships in the Indo-Pacific region to support training of medical practitioners to Australian standards and with pathways to the Australian market.202 As outlined in Chapter 11 – 'Professional and financial services', collaboration between regulatory and professional bodies helps streamline registration processes to facilitate the movement of health and aged care practitioners to fill workforce shortages in Australia and the region. Australia's free trade agreements with the region have specific provisions and platforms for profession-to-profession collaboration, but currently there are no mutual recognition agreements in place for medical professionals between Australia and Southeast Asia.
Such engagement would promote the exchange of knowledge and expertise that would assist in developing best practice in the health sector accreditation and regulatory systems. Governments may be able to play a supportive role, including through development partnerships, in this work. For example, the Katalis program under the Australia–Indonesia Comprehensive Economic Partnership Agreement recently undertook a comparative assessment of nursing standards in Indonesia and Australia to identify gaps in core competencies and support greater mobility for Indonesian nurses.203
Recommendation
- Support state and territory governments to work with Australian institutions to develop a health practitioner training program with key Southeast Asian countries.
Continuing and expanding regulator-to-regulator cooperation, and promoting compatibility between Australian, Southeast Asian and global frameworks and platforms through the TGA regulatory strengthening program, will support health outcomes in the region and its growing health market. Moreover, CSIRO's Australian e-Health Research Centre has world-leading capabilities in digital health and expertise in international digital health standards.
Sharing this expertise with Southeast Asian policy agencies, including supporting the adoption of international digital health standards, will support continued development of the digital health market and cross-border digital health trade. It will also provide a solid foundation for future avenues of cross-border health services trade – for example, clinical decision support utilising specialists in other countries. Both these initiatives could be supported under the Australian Government's recently announced government-to-government partnerships program.
Recommendations
- Expand the TGA regulatory strengthening program to build the capacity of regional regulators.
- Support regional health policy agencies to build digital health capabilities and policy frameworks, led by CSIRO's Australian e-Health Research Centre.
Deepen investment
For medical researchers and startups, cross-border collaboration can accelerate development of new products and treatments, improve access to new markets and open new sources of investment. MTPConnect, Australia's Industry Growth Centre for the medical technology, biotechnology and pharmaceutical sector, is building Australia's connections to global medical innovation. Under MTPConnect's Researcher Exchange and Development within Industry (REDI) fellowship program, Australian researchers, clinicians and professionals have been placed with industry partners in North Asia, the United States and Europe.
The Australian Government could work closely with industry to help accelerate innovation and commercialisation and improve access to markets in Asia through stronger connections with medical innovation hubs in Southeast Asia. Cooperation could include tailoring research and development and product development to address diseases and care needs particular to Southeast Asia through research collaboration and professional exchanges.
Recommendation
- Australian diplomatic missions to work with industry to foster stronger connections between medical innovation hubs in Australia and Southeast Asia.
187 The Institute for Economic and Social Research, Faculty of Economics and Business, Universities Indonesia and Japan International Cooperation Agency, Estimating social infrastructure needs in diverse and dynamic Asia, JICA, 2020, p. 44.
188 United Nations, Department of Economic and Social Affairs, Population Division, Data Portal [dataset], 2022, accessed 5 January 2023.
189 Clearstate, Southeast Asia: The new emerging healthcare market challenge, Economist Intelligence Unit, 2016, p. 12.
190 Fortune Business Insights, 'Southeast Asia Dietary Supplements Market', Fortune Business Insights, 2020.
191 Department of Foreign Affairs and Trade (DFAT), Data analysis using DFATSTARS Database, DFAT, Australian Government, unpublished.
192A Baur, H Yew and M Xin, The future of healthcare in Asia: Digital health ecosystems, McKinsey & Company, 2021, p. 2, accessed 8 March 2023.
193 W Yeung and L Thang, 'Long-Term Care for Older Adults in ASEAN Plus Three: The Roles of Family, Community, and the State in Addressing Unmet Eldercare Needs', Journal of Aging and Health, 2018, 30(10):1499–1515,
194 S Kasumoto, 195 Australian Bureau of Statistics (ABS), Population Projections, Australia, ABS website, 2017, accessed 7 July 2023.
196 National Health and Medical Research Council (NHMRC), NHMRC Engagement in Southeast Asia, NHMRC, Australian Government, Report provided to DFAT, unpublished.
197 Department of Foreign Affairs and Trade (DFAT), Partnerships for a Healthy Region, DFAT website, 23 February 2023, accessed 23 February 2023.
198 Submission from the Medical Technology Association of Australia.
199 World Health Organization (WHO), Nursing and Midwifery Factsheet, WHO, 2022, accessed 3 February 2023. Nurse training data based on Scenario 2: Ageing and graduation as of recent years, World Health Organization, State of the world's nursing 2020: Investing in education, jobs and leadership, WHO, 2020, p. 63.
200 World Health Organization, Global Health Observatory Data Repository [dataset], n.d., accessed 18 December 2022.
201 R Kruk, Independent review of overseas health practitioner regulatory settings. Interim Report, Australian Government, 2023, p. 24.
202 See Recommendation F17 in R Kruk, Independent review of overseas health practitioner regulatory settings. Interim Report, Australian Government, 2023, p. 43.
203 La Trobe University, Comparative Assessment of Nursing Standards in Indonesia and Australia,Indonesia–Australia Comprehensive Economic Partnership Agreement Economic Cooperation Program Katalis, 2022.